
Nitrosamines in Food and Beverages
This is the third in a series of entries examining nitrosamines in a range of products. Our first of two
Accelerate your development process, fulfil marketing authorisations with the right data and strengthen your post-marketing requirements.


Outsourcing Pharmaceutical Analysis is an enabling strategy in today’s business environment. Proficient analytical studies and robust analytical data play a key role in every stage of drug development and Good Manufacturing Practices (cGMP). Addressing typical product development needs, investigating specific concerns around drug safety, supporting complex analysis, and compliance with dynamic regulatory requirements are all SK biotek Ireland Analytical Services’ specialties. Selecting a contract analytical services partner with a rich history of regulatory-compliant testing and development will help you focus on your core business goals.
SK biotek Ireland, based in Swords, Co. Dublin is a contract development and manufacturing organisation (CDMO). We are an SK pharmteco company, leaders in custom chemical development and analysis, API/HPAPI and advanced intermediates manufacturing.





SK biotek Ireland Analytical Services is a global top-tier analytical services provider that prides itself on its entrepreneurial drive, talented workforce and state-of-the-art instrumentation. We partner with you on every step of your journey.

We stay with you throughout your analytical development pathway, focusing precisely on every step but never forgetting the end goal.
We believe in staying agile and flexible in developing the most suitable analytical methods for your needs.
We are continuously developing our people and instrumentation to meet the ever increasing requirements within the industry.
Rather than an arms-length relationship, we work in close collaboration with your scientists to understand avenues already pursued and learn from any experience you have gained, enabling faster and more efficient analytical successes. We have been analyzing some of the world’s most recognized drugs for decades, but embrace the ever changing landscape.
You can be confident we will provide you what you want, when you want it, everytime!

SK biotek Ireland Analytical Services is based in Swords, Co. Dublin – less than 6 kilometres from Dublin Airport – nestled between the US and Continental Europe. Ireland is host to numerous global pharmaceutical companies and manufacturers. Our location enables us to build and grow strong relationships with our customers.

This is the third in a series of entries examining nitrosamines in a range of products. Our first of two
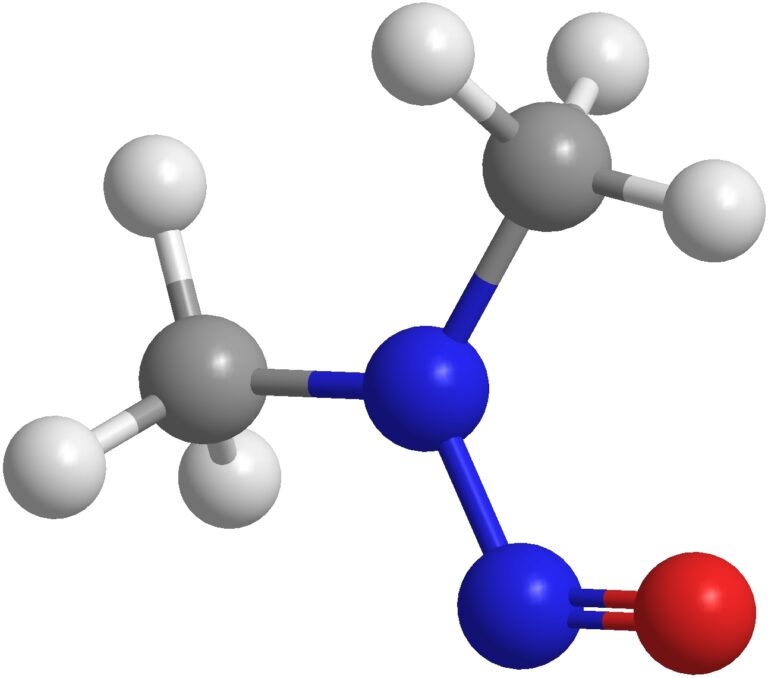
This is the second in a series of entries examining nitrosamines in a range of products. Our first article presented

Our sister company AMPAC Analytical with labs in California and Virginia, has just launched a new blog which will be
We stand by our reputation. Our proven track-record ensures the successful approval of new entities from early stage to commercial launch.
Compliance and health authority regulations are at the forefront of our method development and implementation strategy. For us, a Quality Record is not about just passing every audit by every major regulator from the FDA to PMDA. Instead, it is something that comes as second nature and has become a part of our DNA.
The ‘sense of ownership’ and of ‘being in it together with our customers’ mentality is a proud facet of our culture. It is one of the things that makes us unique.
We work across multiple geographies ensuring open lines of communication, teamwork and multi-disciplinary collaboration, both internally, and externally with our customers.
From your first encounter with our Business Development Team, your dedicated Project Manager, and
every interaction with the teams that come in contact with you, we offer clear lines of communication and regular updates to foster mutual trust.
